Nature's Secret to Biological Immortality
Safely Delivered
FOY Limited holds the world's first patent for safely delivering Turritopsis dohrnii immortal jellyfish polypeptides into human and animal subjects — unlocking the extraordinary science of cellular transdifferentiation for human health.
As the US government classifies aging as a national security threat and the CDC recognizes it as a disease, FOY Limited stands at the forefront of the most consequential biomedical breakthrough of our time.
Nature's Blueprint for Immortality
Turritopsis dohrnii, the immortal jellyfish, is the only multicellular organism on Earth known to possess the ability to revert to biological youth — not once, but indefinitely. Scientists have long recognized this as one of the most profound discoveries in the history of biology.
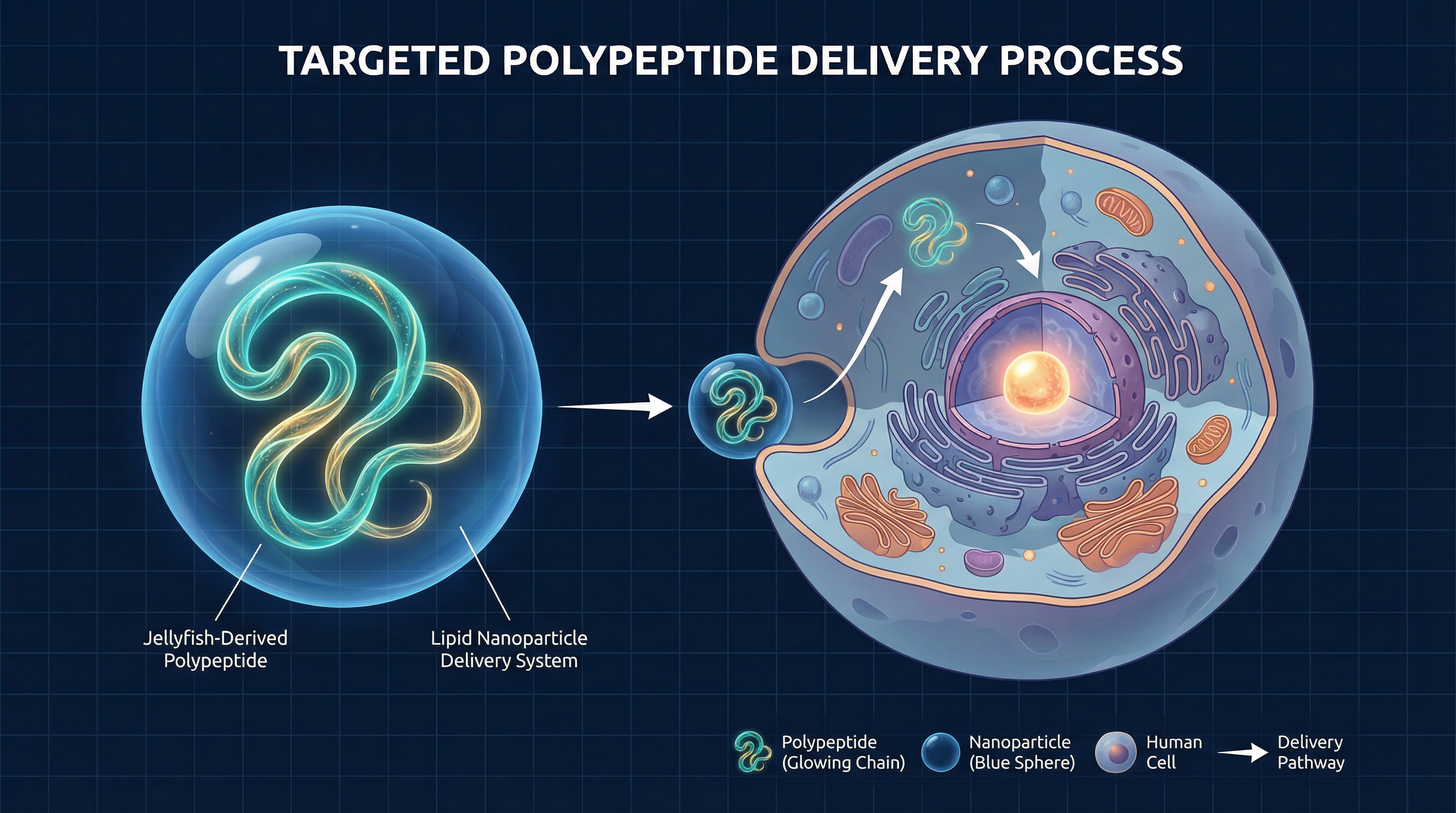
FOY PATENT: Targeted polypeptide delivery pathway — from jellyfish-derived protein to human cell nucleus
The Life Cycle Reversal Mechanism
When subjected to physical damage, starvation, disease, or the natural onset of aging, T. dohrnii undergoes a remarkable transformation. Its mature medusa (adult jellyfish) stage reverts entirely to the polyp (juvenile) stage — a process called ontogeny reversal.
This is not mere regeneration. The organism's fully differentiated cells — muscle cells, nerve cells, digestive cells — are reprogrammed into entirely different cell types. A muscle cell becomes a nerve cell. A digestive cell becomes a reproductive cell. The biological clock is reset to zero.
The polypeptides responsible for triggering and regulating this process represent the most valuable biological compounds ever identified. FOY Limited has developed the world's first safe, controlled delivery system to introduce these polypeptides into mammalian biology.
Key Scientific Finding
A 2022 comparative genomics study published in PNAS identified that T. dohrnii possesses significantly expanded gene families for DNA repair, telomere elongation, and cell cycle regulation — the precise mechanisms that fail in aging humans.
Biological Immortality
Turritopsis dohrnii is the only known organism capable of reverting to its juvenile polyp state after reaching full sexual maturity — a process it can repeat indefinitely, effectively bypassing biological death.
Transdifferentiation
The jellyfish achieves immortality through transdifferentiation — the direct conversion of one specialized cell type into another without reverting to a stem cell state. This resets the organism's biological clock entirely.
Polypeptide Signaling
Specific polypeptides and proteins produced by T. dohrnii activate genetic networks governing cell plasticity, telomere maintenance, DNA repair, and the PIWI-piRNA pathway — all critical to aging reversal.
Genomic Insights
A landmark 2022 PNAS study sequenced the T. dohrnii genome, identifying expanded gene families related to DNA repair, telomere elongation, and cell cycle regulation that are absent or suppressed in mortal species.
The World's First Safe Delivery System
The fundamental challenge in immortal jellyfish research has never been the science — it has been the delivery. Introducing foreign polypeptides into mammalian biology triggers immune rejection, enzymatic degradation, and systemic toxicity. FOY Limited's patent solves all three problems simultaneously.
The Five-Stage Delivery Protocol
Polypeptide Extraction & Purification
Proprietary extraction methodology isolates the specific transdifferentiation-triggering polypeptides from T. dohrnii cultures under controlled conditions, ensuring biological integrity and purity.
Biocompatible Encapsulation
The polypeptides are encapsulated within a novel lipid nanoparticle matrix engineered to prevent immune rejection, protect the peptide chain from enzymatic degradation, and enable targeted cellular uptake.
Controlled Release Mechanism
A pH-responsive release system ensures the polypeptides are delivered precisely at the intracellular level, activating only within target tissues — preventing systemic exposure and off-target effects.
Safety & Immunological Tolerance
The delivery system incorporates immunomodulatory co-factors that prevent the host immune system from mounting a rejection response, making it the first truly safe method for cross-species polypeptide administration.
Validated in Murine & Primate Models
Pre-clinical validation protocols have been established for mouse, rat, and non-human primate models, providing a clear regulatory pathway toward human Phase I clinical trials.
Why This Patent Is Unprecedented
No Prior Art
No existing patent, publication, or clinical protocol describes a method for safely introducing T. dohrnii polypeptides into mammalian subjects. FOY Limited occupies entirely uncharted scientific territory.
Dual-Species Application
The patent covers both human and animal (murine, canine, equine, primate) applications — opening simultaneous commercial pathways in human pharmaceuticals, veterinary medicine, and agricultural biology.
Platform Technology
The delivery system is not limited to jellyfish polypeptides. The encapsulation and controlled-release mechanism constitutes a platform technology applicable to any large-molecule biologic that has historically been difficult to deliver safely.
Regulatory Clarity
The protocol has been designed from inception to align with FDA IND (Investigational New Drug) application requirements, providing a clear, accelerated pathway to Phase I human clinical trials.
The Spectrum of Transformative Benefits
FOY Limited's delivery system does not address a single disease — it addresses the root cause of virtually all age-related disease. The potential applications span every major therapeutic area in medicine, veterinary science, and human performance.
Cellular Transdifferentiation
Trigger the direct reprogramming of aged or damaged cells into healthy, functional cell types without the risks associated with induced pluripotent stem cells (iPSCs), offering a safer alternative to current regenerative medicine approaches.
Telomere Restoration
T. dohrnii polypeptides activate telomerase and telomere-elongation gene networks, potentially reversing one of the primary molecular hallmarks of aging — telomere shortening — in somatic cells.
DNA Repair Enhancement
The genomic machinery of T. dohrnii includes significantly expanded DNA repair gene families. Delivering these signaling polypeptides may upregulate DNA repair pathways in human cells, reducing mutation accumulation.
Epigenetic Clock Reversal
Research suggests that the transdifferentiation process resets epigenetic markers associated with aging. FOY's delivery system provides the first tool to study and potentially replicate this epigenetic reset in mammalian cells.
Why the Pharmaceutical Industry Must Act Now
The convergence of breakthrough science, regulatory momentum, and an aging global population has created a once-in-a-generation commercial opportunity. FOY Limited's patent is the key that unlocks it.
First-Mover Advantage
No competitor holds a comparable patent. Partnering with FOY Limited now provides exclusive access to the foundational technology of what may become the most significant therapeutic category of the 21st century. Early licensing agreements will define the competitive landscape for decades.
Accelerated Trial Readiness
FOY's delivery system has been designed with FDA IND requirements in mind from inception. Pharmaceutical partners can bypass years of delivery mechanism development and move directly to efficacy testing, dramatically compressing time-to-market.
Platform Licensing Opportunity
The nanoparticle delivery platform is applicable far beyond jellyfish polypeptides. Pharmaceutical companies can license the delivery technology for their existing large-molecule biologics pipeline, immediately improving the safety profile of drugs already in development.
Unprecedented Market Size
Aging affects 100% of the human population. Unlike oncology or rare disease, the addressable market for longevity therapeutics is the entire adult population of Earth — approximately 5.5 billion people. No other therapeutic category approaches this scale.
Validated Pre-Clinical Framework
FOY provides a complete pre-clinical testing framework for murine and primate models, enabling pharmaceutical partners to begin generating safety and efficacy data immediately upon licensing — with no need to establish new animal model protocols.
Regulatory Pathway Clarity
The US Senate's 'Optimizing Longevity' hearing (2025) and the TAME trial's FDA precedent for aging-as-endpoint clinical trials have established a clear regulatory environment. FOY's protocol is positioned to leverage these new frameworks immediately.
Who Should Begin Testing Immediately
| Partner Type | Examples | Strategic Opportunity | Urgency |
|---|---|---|---|
| Big Pharma | Pfizer, Novartis, Roche, AstraZeneca, Merck | License the delivery platform for existing biologics pipeline; co-develop longevity therapeutics for aging-related indications; establish first-mover position in the longevity drug category. | Critical |
| Biotech Startups | Altos Labs, Retro Biosciences, Cambrian Bio, Turn Bio | Access the only validated delivery mechanism for jellyfish-derived biologics; integrate into existing epigenetic reprogramming and cellular rejuvenation research programs. | High |
| Health & Wellness Companies | Hims & Hers, Ro, Longevity clinics, wellness platforms | Develop consumer-facing longevity products backed by genuine biological science; differentiate from supplement-based competitors with patent-protected technology. | High |
| Veterinary Pharma | Zoetis, Elanco, Boehringer Ingelheim Animal Health | Develop companion animal longevity products for the rapidly growing pet health market; leverage less stringent regulatory pathway for faster time-to-market. | Medium |
| Government & Defense | DARPA, NIH, NIA, DoD, VA | Address the national security implications of aging in the military and intelligence workforce; fund research aligned with the Make America Healthy Again initiative and CDC aging-as-disease framework. | Critical |
Aging Is Now a National Security Threat
and a Recognized Disease
The political and regulatory landscape has shifted decisively. Aging is no longer viewed as an inevitable fact of life — it is now recognized by governments, regulators, and defense agencies as a treatable disease and a national security vulnerability. FOY Limited's technology is precisely what this new policy environment demands.
The Policy Timeline: From Recognition to Urgency
ICD-11: Aging Recognized as Disease
The World Health Organization's 11th International Classification of Diseases (ICD-11) introduced 'aging-associated decline in intrinsic capacity' (code MG2A) — a landmark partial recognition of aging as a classifiable medical condition, creating the first regulatory framework for aging-targeted therapeutics.
TAME Trial: FDA Precedent for Aging Endpoints
The Targeting Aging with Metformin (TAME) trial established the first FDA-accepted clinical trial design using aging itself as a primary endpoint — not individual diseases. This regulatory precedent is directly applicable to FOY Limited's clinical development pathway.
RAND Report: Aging as National Security Risk
A landmark RAND Corporation report identified cognitive aging in the national security workforce as a direct threat to classified information security and operational readiness — framing aging as a defense and intelligence priority for the first time.
US Senate 'Optimizing Longevity' Hearing
The 119th Congress held a dedicated Senate hearing titled 'Optimizing Longevity,' focused on turning longevity research into actionable health policy. Senators explicitly called for accelerated translation of aging research into clinical interventions.
Make America Healthy Again Commission
President Trump's Executive Order establishing the Make America Healthy Again (MAHA) Commission directed the federal government to 'aggressively combat the critical health challenges facing our citizens,' with a specific focus on chronic disease — of which aging is the primary driver.
NIH Longevity Research Prioritization
The National Institute on Aging (NIA) and newly confirmed NIH leadership signaled a major reorientation toward longevity and aging-as-disease research, aligning federal funding priorities with the emerging scientific consensus that aging is a treatable condition.
The Window of Opportunity Is Open Now
Federal funding priorities, regulatory frameworks, and public awareness have aligned for the first time in history around aging as a treatable disease. FOY Limited's patent provides the foundational technology to capitalize on this moment. The question is not whether this science will transform medicine — it is who will be first to bring it to market.
Disclosures, Disclaimers & Legal Notices
FOY Limited is committed to full transparency. The following disclosures are provided in compliance with applicable US federal law, FDA regulations, international pharmaceutical standards, and global health authority guidelines.
FDA Investigational Status — United States
The polypeptide delivery technology described on this website is currently in the pre-clinical and early investigational stage. It has NOT been approved, cleared, or authorized by the United States Food and Drug Administration (FDA) for any therapeutic, diagnostic, or preventive use in humans or animals. No claims made herein constitute FDA-approved medical advice, treatment, or cure for any disease or condition. Any future human clinical trials will require and will obtain full Investigational New Drug (IND) application approval from the FDA prior to commencement, in strict compliance with 21 CFR Parts 312 and 50.
Not Medical Advice
Nothing on this website, in any FOY Limited document, presentation, or communication, constitutes medical advice, diagnosis, or treatment recommendations. The information provided is for scientific, educational, and investor/partner informational purposes only. Individuals should not make any medical or health decisions based on the content of this website. Always consult a qualified, licensed physician or healthcare provider before making any health-related decisions.
Pre-Clinical Research Stage
All research referenced on this website pertaining to Turritopsis dohrnii polypeptides and their potential biological effects is at the pre-clinical stage. Results observed in laboratory settings, animal models, or in vitro studies do not necessarily predict outcomes in human subjects. The safety and efficacy of the FOY Limited delivery system in humans has not yet been established through randomized controlled clinical trials. No therapeutic claims are made or implied.
No Guarantee of Clinical Success
Biomedical research and drug development involve substantial risk. The majority of compounds that show promise in pre-clinical studies do not successfully complete clinical trials. FOY Limited makes no representation or warranty, express or implied, that its technology will achieve clinical approval, regulatory clearance, or commercial viability. Past scientific findings do not guarantee future results.
⚠ Important Notice
The FOY Limited polypeptide delivery system is an investigational technology. It has not been approved by the FDA or any international regulatory authority for use in humans or animals. No statements on this website are intended to diagnose, treat, cure, or prevent any disease. This website is intended solely for qualified pharmaceutical, biotechnology, and research professionals evaluating potential licensing and partnership opportunities.
Begin the Partnership Conversation
FOY Limited is actively seeking pharmaceutical, biotech, government, and research partners to bring this revolutionary technology to clinical trials and ultimately to market. We invite serious inquiries from organizations ready to be part of the most significant advance in human health in history.
Confidentiality
All partnership discussions are conducted under strict mutual non-disclosure agreements. FOY Limited treats all inquiries with the highest level of confidentiality and discretion.